You might think cancer clinical trials are just lab experiments far removed from your hospital visits. But here’s the truth: they’re life-or-death pathways where new treatments get tested, and participants often gain early access to therapies standard care hasn’t caught up with yet. In 2023 alone, around 4,000 such trials ran globally-each following strict rules to balance hope with safety.
Imagine you’ve exhausted standard treatments. A Phase II trial for immunotherapy might shrink tumors when nothing else works. Real patients live through this. Take 'SurvivorStrong,' whose melanoma vanished after joining a trial. But joining isn’t simple. Strict eligibility rules exclude most people, and traveling weeks can disrupt daily life. This guide cuts through confusion: what each phase does, who qualifies, and how to decide if a trial fits your situation.
The Five Stages of Cancer Clinical Trials
Clinical trials don’t rush straight to testing new drugs. They move like stepping stones, each stage answering critical questions before risking larger groups. Here’s how they break down:
| Phase | Participants | Main Goal | Duration |
|---|---|---|---|
| Phase 0 | 10-15 | Test if drug reaches cancer cells | Few weeks |
| Phase I | 15-80 | Assess safety & dosage limits | Months to years |
| Phase II | 25-100 | Check effectiveness against specific cancers | 6-12 months |
| Phase III | 100s-1000s | Compare new treatment vs standard care | 1-4 years |
| Phase IV | Thousands | Monitor long-term safety post-approval | Years to decades |
Phase 0 trials are like tiny tests-using subtherapeutic doses to see how cancer cells react without full treatment effects. Think of it as a preliminary reconnaissance mission. By Phase I, researchers push higher doses carefully. Imagine being among the first 20 volunteers trying a novel oral drug for pancreatic cancer. Your blood work gets checked obsessively, side effects tracked hourly. This phase isn’t about curing anyone yet-it’s about figuring out safe dosage ranges.
Once safety clears, Phase II ramps up intensity. Now doctors ask: Does this actually fight *your* cancer type? If you have stage 3 lung cancer and join a trial testing a targeted therapy, scans every few weeks measure tumor shrinkage. Success rates here vary wildly. Some trials halt because unexpected toxicities emerge-even if earlier stages looked promising.
Phase III becomes high-stakes chess. You might be randomly assigned either the experimental treatment or best current care. Why randomization? To eliminate bias. Suppose half the trial arms receive checkpoint inhibitor combos while others get chemo. After analyzing thousands of outcomes, scientists declare winners. These results determine whether governments approve new drugs nationwide.
Don’t forget Phase IV-the silent guardian. Even after approval, monitoring continues. Rare side effects showing up years later? That’s Phase IV’s job. Consider Kevadar, an older leukemia drug pulled in 2024 due to heart risks detected only after large-scale post-market studies.
Who Qualifies-and Who Gets Left Behind?
Hear this stark fact: roughly 8 out of 10 cancer patients fail eligibility checks. Why? Trials demand narrow criteria. For example, a breast cancer study might require:
- Specific genetic markers (like HER2 positivity)
- No prior brain metastases
- Limited exposure to previous chemotherapy types
This filters out elderly patients with multiple health issues. Diversity gaps persist too-only 8% of U.S. trial participants identify as Black despite comprising 13% of cases. Researchers increasingly recognize this skews results. Imagine testing prostate cancer therapies predominantly on white males; findings may not reflect African American experiences where incidence rates differ significantly.
Patient navigators now bridge these gaps. At centers like Queensland’s Royal Brisbane Hospital, dedicated staff help decode medical jargon during pre-trial consultations. They clarify terms like "placebo control"-where some participants receive inactive substances instead of active treatments-to ease fears about receiving substandard care.
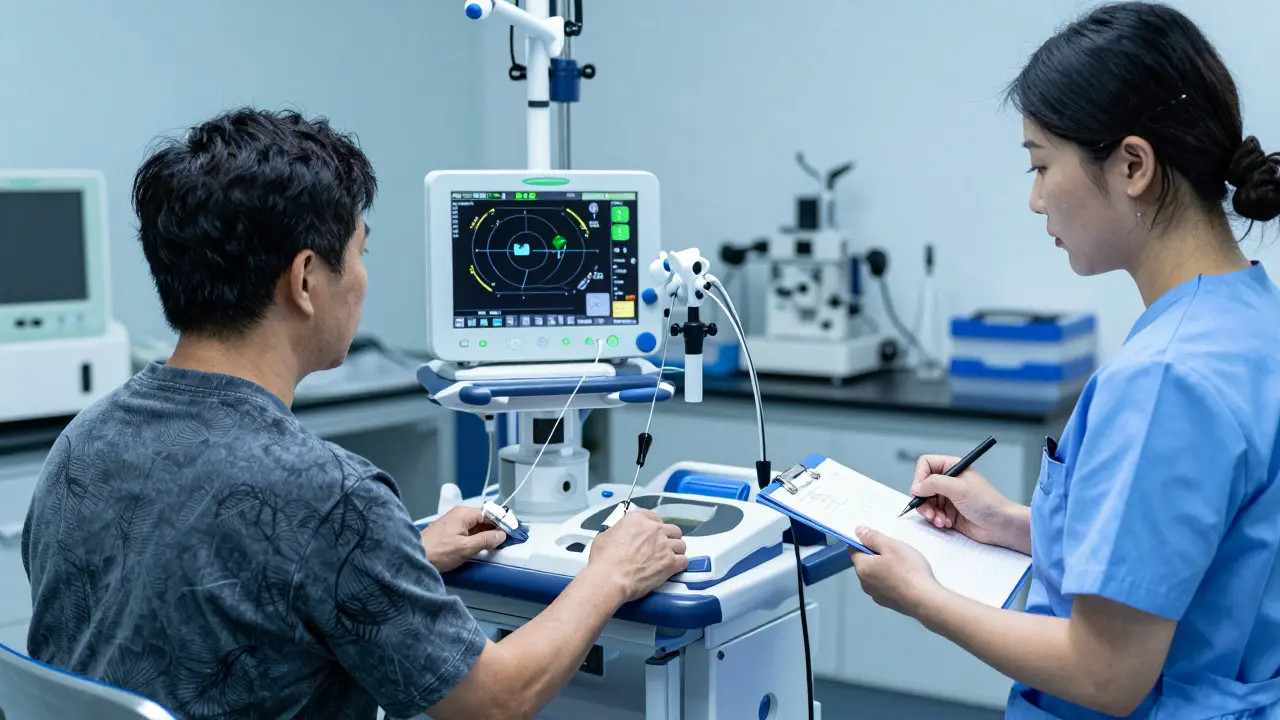
Real Benefits Beyond New Treatments
Beyond accessing cutting-edge science, many report deeper engagement with their healthcare team. During my coverage of Western Australia’s trials last year, one ovarian cancer survivor shared:
"In standard care, appointments were rushed. In the trial, coordinators explained every scan result. I felt heard."
This heightened oversight catches complications faster. One participant discovered kidney issues during routine trial bloodwork that otherwise might have gone unnoticed until crisis point.
Yet logistics bite hard. Transportation costs drain savings accounts. Rural residents in WA face hour-long drives weekly-compounding fatigue from aggressive treatments. New hybrid models address this via remote monitoring wearables. Picture tracking vital signs from home apps rather than clinic visits. Such innovations aim to democratize access without sacrificing data quality.

Weighing Risks Against Potential Rewards
All trials carry uncertainty. Half of Phase II initiatives collapse before reaching broader testing due to insufficient efficacy or dangerous side effects. Still, consider this: approximately 70% of approved cancer drugs trace back to successful multi-center studies conducted over past decades. Each failure teaches future developers pitfalls to avoid-a crucial debt repaid by earlier participants.
Randomization anxiety plagues hopeful candidates worried about missing out on breakthrough options. Remember: modern ethical frameworks ensure control groups transition to proven therapies once superiority emerges. Plus, open-label extensions allow continued access after primary endpoints finish. Transparency remains non-negotiable-ethical review boards scrutinize informed consent processes continuously.
Steps to Evaluate Your Fit
Ready to explore possibilities? Follow this roadmap:
- Schedule initial consultation: Most major centers reserve same-week slots for interest interviews. Bring pathology reports highlighting molecular profiles.
- Review inclusion/exclusion lists: Average trials contain ~28 criteria; cross-check thoroughly with oncologist input.
- Contact patient support networks: Organizations like Cancer Council WA provide navigator referrals.
- Discuss financial implications: Confirm insurance coverage for required procedures beyond standard care costs.
- Set expectations early: Clarify travel reimbursement policies upfront; many sponsors cover airfare/hotel stays.
Time investment looms large too. Screening typically consumes two weeks between application and final enrollment confirmation. Factor this into decision-making timelines, especially for rapidly progressing diseases requiring swift action.
Can I drop out of a clinical trial anytime?
Yes-you retain full autonomy throughout participation. Simply inform the coordinator; withdrawal doesn’t affect ongoing standard care access.
Do placebo controls ever receive active treatment later?
Often yes. Cross-over designs permit switching upon demonstrating benefit, preserving scientific integrity while ensuring equitable care.
How do I find locally available trials?
Search national registries like Therapeutic Goods Administration databases filtered by location/disease subtype. Consult hospital liaison officers familiar with regional programs.
Are pediatric cancer trials different from adult studies?
Pediatric protocols adjust dosing calculations based on weight/surface area metrics unique to growing bodies. Consent involves dual-parental authorization alongside age-appropriate child assent.
What happens if unexpected toxicity arises mid-trial?
Immediate protocol activation halts administration until independent reviews assess causality severity. Participants shift to palliative management aligned with established guidelines.







Dan Stoof March 30, 2026
Wow this is absolutely amazing stuff!!! So many people give up hope way too early which is sad!!! It gives me such good feels to see science working hard for us!!! We really do have so much to look forward to with these trial phases!!! Every single step matters so much!!! Keep the faith strong everyone!!!
Calvin H March 30, 2026
Sure let’s bet our lives on lab rats before we try living normally.
Katie Riston April 1, 2026
It is truly fascinating how society views medical intervention today.
Many people fear the unknown without understanding the process deeply.
We often forget that progress requires sacrifice from those brave enough to participate.
The moral weight placed on clinical trial participants is immense and rarely acknowledged properly.
One must consider the societal obligation to advance knowledge even when personal risk exists.
Safety protocols exist not just for the individual but for the collective future of humanity.
Without volunteers in phase one studies we would remain stagnant in treatment options forever.
The randomness involved in control groups creates its own ethical dilemma regarding choice.
Sometimes the best care available right now is still inferior to what could be discovered tomorrow.
Patients navigating this landscape deserve better support systems than currently offered globally.
Insurance coverage gaps create barriers that prevent necessary data collection across demographics.
Diversity in testing populations remains a critical issue that demands immediate attention from regulators.
We cannot afford to ignore the disparities highlighted in recent statistics about minority enrollment.
Transparency in reporting outcomes ensures trust remains intact between researchers and the public community.
Ultimately every decision involves weighing potential loss against theoretical gain in complex scenarios.
Cameron Redic April 2, 2026
You sound like you’re trying to write a thesis instead of reading a blog post.
Most people don’t have time for all that fluff here.
Just read the facts listed in the table and move on quickly.
Efficiency matters more than philosophy in medical contexts.
Michael Kinkoph April 3, 2026
Your lack of depth is absolutely staggering!!!
How dare you dismiss deep analysis as mere fluff!!!
The nuance matters immensely!!!
Ignorance is never an acceptable excuse in serious discussions!!!
Brian Yap April 4, 2026
Down here in Oz we deal with similar things at the local hospitals sometimes.
It’s pretty wild seeing the numbers on eligibility requirements honestly.
Cheers for laying it out clearly mate.
Ruth Wambui April 5, 2026
Hospitals always hide the truth about side effects behind bureaucracy walls.
They want your data more than they want you healthy actually.
Big pharma profits drive these studies far more than patient safety does.
William Rhodes April 6, 2026
Stop spreading negativity about the system that saves lives daily!!!
Scientists work tirelessly to help us survive against all odds!!!
Doubt helps nobody in their darkest hour!!!
sanatan kaushik April 8, 2026
People talk too much about risks.
Just go for it if doctor says yes.
Waiting is worse than trying something new.
Biraju Shah April 9, 2026
I agree waiting is bad but taking advice carefully is important too.
Balance keeps everyone safe while moving forward.
Marwood Construction April 10, 2026
The statistical breakdown of participant numbers per phase provides valuable context for potential candidates.
Understanding the duration expectations prevents logistical issues later on during treatment.